|
||||||||||||||||||||||||||||||||||
To unsubscribe, click here
|
||||||||||||||||||||||||||||||||||
[Paper: Mean-field and linear regime approach to magnetic hyperthermia of core-shell nanoparticles: can tiny nanostructures fight cancer? Marcus S. Carrião, Andris F. Bakuzis. Nanoscale, 2016,8, 8363-8377. DOI: 10.1039/C5NR09093H]

Hyperthermia, for cancer treatment, is induced by increasing the temperature to activate tumor cell death. This high temperature can be created by introducing nanoparticles into the tumor that function as heaters, and after their function is completed they are eliminated by the body. Magnetic nanoparticles may be used in these treatments because they have the ability to generate heat when subjected to alternating magnetic fields of specific intensity and frequency.
A work on nanomedicine (nanotechnology used in medicine) fully conducted at the Brazilian Federal University of Goiás (UFG) suggests a new strategy using hyperthermia for cancer treatment: using smaller magnetic nanoparticles than those normally used and composed of more than one material, which could provide several advantages to the patient. To reach this conclusion, the researchers developed an innovative theoretical method that paves the way for manufacturing magnetic nanoparticles optimized for hyperthermia. The study was reported in a paper published in the prestigious Nanoscale journal, signed by the doctoral student Marcus Carrião dos Santos and his supervisor Andris Figueiroa Bakuzis, professor at the Physics Institute of UFG.
Hyperthermia cancer treatment generally uses nanoparticles that are relatively large (20 nm size range) and homogeneous (from a single material), which are considered as the most effective to generate heat according to theoretical studies based on traditional methods. However, these “large” nanoparticles accumulate quickly in the liver and it may take several months or years for these particles to leave the body of the patient being treated. On the other hand, nanoparticles smaller than 10 nm are rapidly eliminated in the urine, reducing the possibilities of intoxication and thereby increasing the selection of materials that can be used to manufacture them.
The relationship between particle size and excretion route (liver or kidney) was a conclusion reached by Bakuzis and colleagues from evidence reported in the scientific literature and pre-clinical studies (in vivo) carried out within a multidisciplinary research network, coordinated by Bakuzis, and aimed at solving problems associated with the use of magnetic nanoparticles for cancer treatment.
In addition, smaller nanoparticles have better distribution and penetration in tumors, among other advantages in the context of cancer treatment.
Aware of these characteristics, Bakuzis and dos Santos investigated the possibility of manufacturing nanoparticles of less than 10 nm that could efficiently generate heat. An important insight came from an article published in 2011 in the Nature Nanotechnology journal (Nat. Nanotech. 6, 418 (2011)). Professor Bakuzis says that “in this article, the researchers concluded experimentally that certain heterogeneous (from different materials) core-shell structures of spinel ferrites warmed more efficiently than homogeneous particles”.
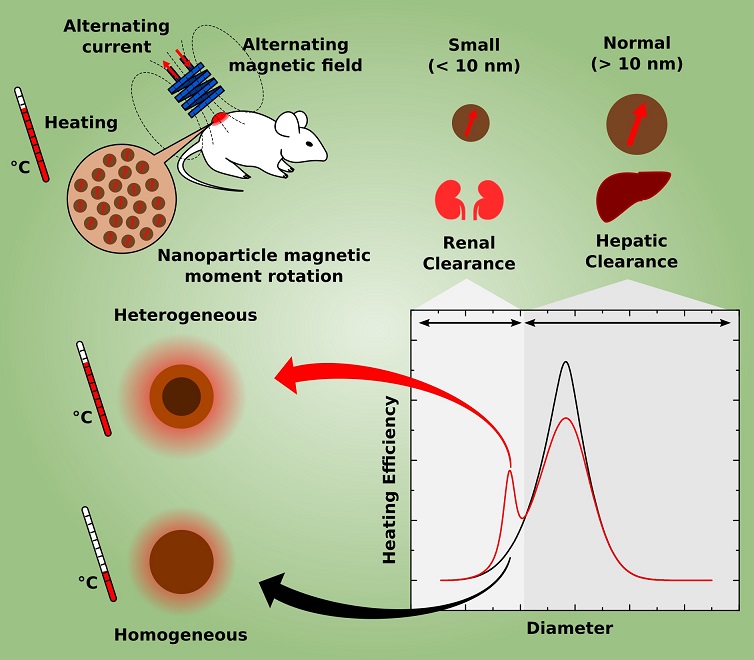
The pair of scientists then decided to theoretically investigate whether nanoparticles less than 10 nm formed by one material core and a shell of another material could efficiently generate heat and how to optimize them for this function. However, the conventional methods available for this modeling were not adequate. In fact, they considered the nanoparticle as a homogeneous entity, ignoring the fact that the surface atoms and the core atoms respond differently to the application of a magnetic field. This oversight became more significant in the study of particularly heterogeneous particles such as those they intended to study, the reason why the researchers from Goiás decided to develop a more suitable model for the study object. Bakuzis explains that “in the paper we presented the first analytical hyperthermia model of core-shell nanoparticles within the linear response and mean-field theory, and from these calculations we pointed out important materials properties to achieve efficient heat generation.”
The results obtained by the physicists and published in the paper may have a significant impact in a health issue that concerns humanity, cancer cure. “Our studies indicate that it is possible to develop small particles for cancer treatment that can be quickly eliminated from the body via the kidneys. In particular, by combining different materials in the nanostructure”, summarizes Bakuzis.
To work with impact on this interface theme, Bakuzis is always in contact with a pool of knowledge of various areas. In addition to leading the multidisciplinary nanomedicine network that includes researchers with backgrounds in tumor biology, genetics, physiology, pharmacy, veterinary medicine, biophysics, physics, medical physics and chemistry, the professor and his group actively participate in scientific events that bring together many different professionals, including doctors with various specializations already using hyperthermia in humans for cancer treatment. “These scientific contacts are fundamental in interface areas such as the one our group works with,” concludes Bakuzis.
The research that led to the paper in the Nanoscale journal received funding from the Brazilian National Scientific and Technological Development Council (CNPq) and from the Research Foundation of the State of Goiás (FAPEG) and was carried out as part of the doctoral work of Marcus Carrião dos Santos.
[Paper: Charge transfer effects on the chemical reactivity of PdxCu1−x nanoalloys. M. V. Castegnaro, A. Gorgeski, B. Balke, M. C. M. Alves and J. Morais. Nanoscale, 2016,8, 641-647. DOI: 10.1039/C5NR06685A]
Taming the reactivity of nanoalloys
When, in 2009, the Electron Spectroscopy Laboratory (LEe-) group of the Federal University of Rio Grande do Sul (UFRGS) decided to start developing in-house metal nanoparticles required for their studies, they came across some issues. Many synthesis methods reported in the literature did not provide the expected results when made in the Brazilian laboratory.

Strongly motivated by curiosity, as usual, says professor Jonder Morais, LEe- researcher, the group members were able, after much dedication, to develop new routes of synthesis that, in addition to being reproducible, are environment-friendly, efficient and cost-effective. “The first articles were published in international journals in 2013, initially with palladium (Pd), platinum (Pt) and silver (Ag) nanoparticles applied to the catalytic decomposition of nitric oxide. Subsequently, we published some works focused in “in situ” studies aimed at determining the mechanisms of formation and growth of monometallic nanoparticles. We have recently started reporting the results obtained with more complex systems, such as palladium and copper (Pd-Cu) nanoalloys,” states Professor Morais.
The latter group includes the results recently reported in an article published in the journal Nanoscale, whose main authors are Professor Jonder Morais and Marcus Vinicius Castegnaro, a physics doctoral student at UFRGS, advised by Morais. The research covered the entire process from the production of nanomaterials to the survey of their applications. “It was important to have dedicated students, willing to face the challenge of preparing accurately their own samples, and correlating the electronic and structural properties to understand the final properties in terms of chemical reactivity,” says Morais.
In the article published in Nanoscale, nanoparticles composed of palladium and copper alloys were produced by applying a simple method developed by the LEe- team. This process is carried out under mild conditions to the environment and health (aqueous, ambient temperature and pressure, and use of cheap and innocuous substances, such as ascorbic acid and sodium citrate). Several samples were synthesized by this route, containing three different amounts of palladium and copper atoms.
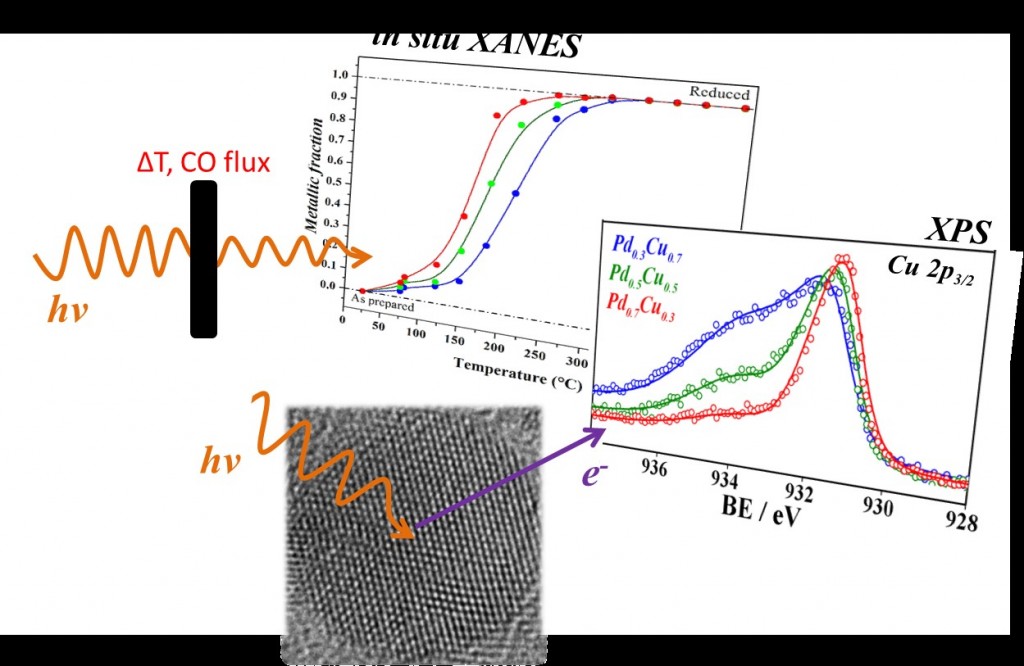
The synthesized nanoparticles have undergone a series of analyses conducted at UFRGS, in Porto Alegre (Rio Grande do Sul State), they traveled to Campinas (São Paulo State) for another series of analyses on equipment of the National Center for Research in Energy and Materials (CNPEM) and crossed the ocean to Johannes Gutenberg University, in Germany, for some additional measures. From characterization, the authors concluded that the nanoparticles were approximately 4 nm in size and were highly crystalline, among other characteristics. In addition, through experiments conducted by the XANES in situ technique, the team of scientists exposed the nanoparticles to carbon monoxide (CO) at 450 ° C and surveyed the reactivity of the nanoalloys, i.e., their ability to react chemically.
After studying the results of the characterization, the authors of the article were able to conclude that the alloy composition affects the ability of nanoalloys to reduce (gain electrons) and to oxidize (lose electrons). In fact, the greater the amount of palladium, the easier the reduction, and the harder the oxidation.
“The published results, obtained by the association of several experimental techniques are relevant to an understanding of the origin of high catalytic reactivity of palladium and copper (Pd-Cu) nanoalloys, as well as to elucidating similar behavior of other bimetallic systems”, highlights Jonder Morais. “Mostly, these results can be used in the “design “of new nanomaterials more efficient for various applications, such as in the petrochemical industry, in fuel cells or in the control of greenhouse gas emissions,” he concludes.
The scientific paper by members of the Brazilian community on Materials research featured this month is:
Luís F. da Silva, Ariadne C. Catto, Waldir Avansi, Laécio S. Cavalcante, Juan Andrés, Khalifa Aguir, Valmor R. Mastelaro and Elson Longo. A novel ozone gas sensor based on one-dimensional (1D) α-Ag2WO4 nanostructures. Nanoscale (Print), 2014, v. 1, p. 1-2. DOI: 10.1039/C3NR05837A
New ozone sensor based on nanorods of silver tungstate
A study carried out by a research group from Brazil, with collaboration from French and Spanish scientists, has reported, for the first time, gas detection properties in nanorods of silver tungstate in its alpha phase (α-Ag2WO4).The study showed that this material can be applied as a resistive sensor, displaying great performance when detecting ozone (O3). The work was and coordinated by Elson Longo, Professor at the São Paulo State University “Júlio de Mesquita Filho” (UNESP).
Resistive gas sensors are basically built from a material capable of changing its electrical properties when molecules of a certain gas are adsorbed in its surface. In the specific case of the silver tungstate, when it is submitted to an oxidant gas such as ozone, there is an increase in its electrical resistance that is proportional to the presence and concentration of the gas.
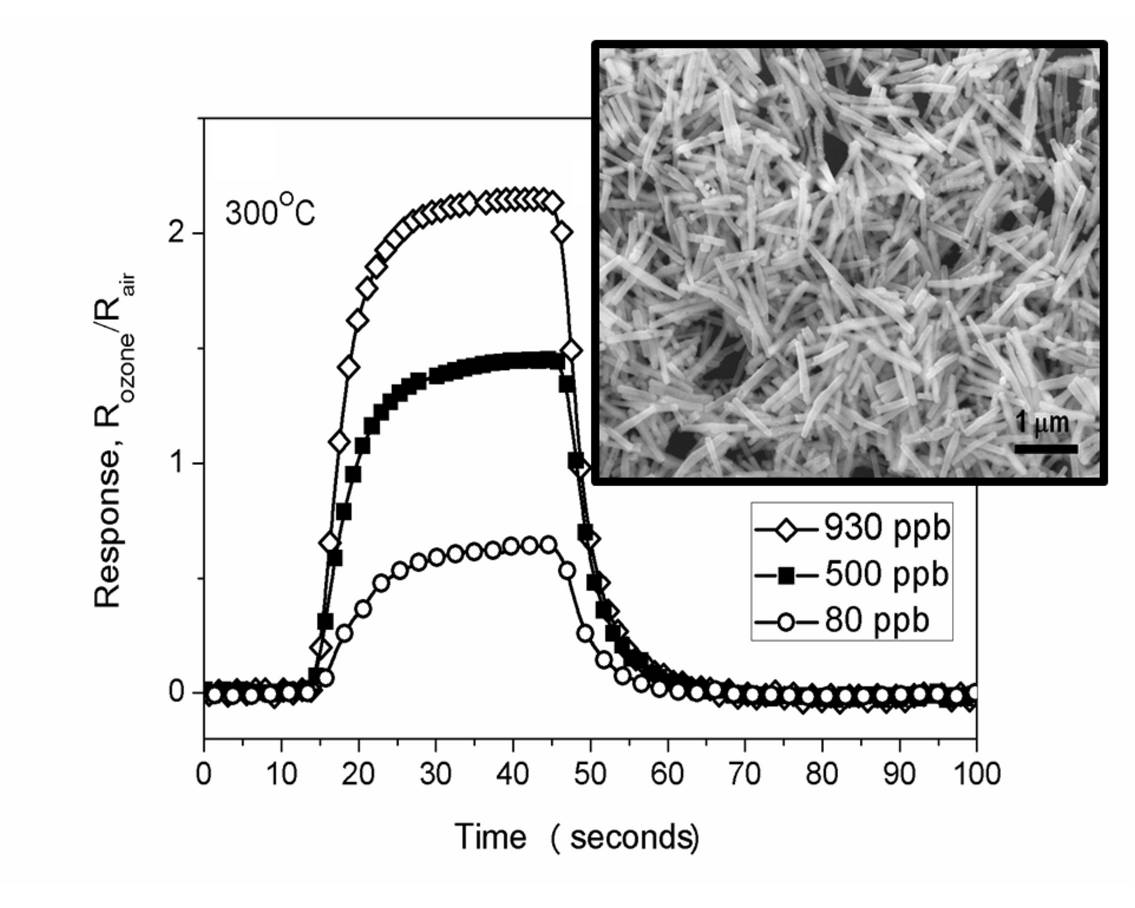
In this work, the Brazilian scientists synthesized nanorods of silver tungstate and assembled a sensor based on such nanoparticles. They had put the sensor in a temperature controlled test chamber, exposed it to different concentrations of ozone gas, from 80 to 930 parts per billion (ppb), and evaluated its capacity to detect the ozone.
Present in high atmospheric layers, the ozone plays an important role protecting living beings by absorbing the solar ultraviolet radiation. The ozone is also used by people in several applications, such as, for example, cleaning water. However, the exposition to the gas in certain concentrations may lead to health issues as headache, burning and irritation in the eyes, and respiratory system problems. The World Health Organization (WHO) recommends avoiding the exposure to ozone gas above 120 ppb.
“Subjecting the compound to low amounts of ozone, we observed a fast response, as well as a very short recovering time, making its properties comparable or even better than traditional sensors as tin dioxide (SnO2), tungsten trioxide (WO3), and indium oxide (In2O3),” says Luís Fernando da Silva, first author of the article and postdoctoral fellow of the São Paulo Research Foundation (Fapesp) at UNESP’s Chemistry Institute of Araraquara.
The results were published online in the peer reviewed journal Nanoscale in the end of January this year.
Background of the paper
The studies with silver tungstate started in the postdoctoral research of Laécio Cavalcante, currently a Professor at Piauí State University (UESPI). Cavalcante synthesized nanorods of silver tungstate using microwave-assisted hydrothermal technique (process that was also used in the synthesis of nanorods from the paper published by Nanoscale). Performing electronic microscopic analysis with the microscope of the Chemistry Institute of Araraquara, the group of scientists coordinated by Professor Longo noticed that the interaction of the electron beam with the material was stimulating the growth of metallic silver particles on the surface of the nanorods. The result of this paper led to an article published in April last year by Scientific Reports (DOI: 10.1038/srep01676).
“Since then, Professor Elson Longo has researched and encouraged the investigation of the potentiality of the α-Ag2WO4 compound”, comments Luís Fernando da Silva. Longo, his team and partners have already observed that the material holds bactericidal (J. Phys. Chem. A, 2014; Doi:10.1021/jp410564p), photoluminescent (J. Phys. Chem. C, 2014, DOI: 10.1021/jp408167v), and photocatalytic properties, with a series of possible applications.
“Based on these potential applications”, Luís Fernando da Silva adds, “I, Professor Waltir Avansi Junior from the Physics Department of the Federal University of São Carlos (UFSCar), jointly with Professor Valmor Mastelaro from the São Carlos Physics Institute of São Paulo University (USP) and his PhD student, Ariadne Catto, started investigating about the detection properties of the non-irradiated α-Ag2WO4 compound (without nanoparticles of metallic silver)”. During the experiments, Silva says, the team noticed that the material was sensitive to detect ethanol and acetone steam and, ultimately, ozone gas, even in low amounts. Assisted by Professors Khalifa Aguir from Université Aix-Marseille (Marseille, France), and Juan Andrés, from Universitat Jaume I (Castelló, Spain), they prepared the communication published by Nanoscale, a renowned journal in the nanotechnology field.
The studies concerning silver tungstate performed by the team of Professor Longo might not end there. According to Luís Fernando da Silva, the team will assess the capacities of the material to detect other gases. In addition to that, returning to the silver tungstate nanorods with nanoparticles of metallic silver, the scientists are going to study the effects of electron irradiation on the gas detection capacity of the material.
“This paper contributes to the discovering of new materials applied as gas sensors”, states the postdoctoral fellow. “However, complementary examinations are necessary in order to achieve a deeper comprehension of the mechanisms involved in the detection, adsorption and desorption processes of the gas(es)”, he concludes.