[Paper: Giant Barocaloric Effects in Natural Rubber: A Relevant Step toward Solid-State Cooling. N. M. Bom, W. Imamura, E. O. Usuda, L. S. Paixão, and A. M. G. Carvalho. ACS Macro Lett. 2018, 7, 31-36. dx.doi.org/10.1021/acsmacrolett.7b00744]
Rubber under Pressure for Solid-State Cooling
A team of researchers from Brazil has found that vulcanized natural rubber prevails over any other material already studied in its capacity to change temperature by being compressed and decompressed – a phenomenon known as “barocaloric effect.”
The discovery opens up interesting possibilities of using vulcanized natural rubber in advanced applications, especially in the area of “solid-state cooling.” This term refers to refrigeration systems (such as refrigerators or air conditioners) that are based on the use of solid state refrigerant materials to absorb the heat of the system to be cooled and transferred to an external environment. Conventional devices use fluids (gaseous and liquid states). The research was reported in an article recently published in ACS Macro Letters, a journal of the American Chemical Society publisher in the field of Polymer Science and the related matters, whose impact factor is 6,185.
“Since natural rubber heats up when pressed (more than 20 degrees above the initial temperature) and cools when the pressure is released (at least 20 degrees below the initial temperature), we believe it can be used as refrigerant material in a refrigerator,” explains Alexandre Magnus Gomes Carvalho, researcher at the Brazilian Synchrotron Light Laboratory (LNLS) and corresponding author of the article.
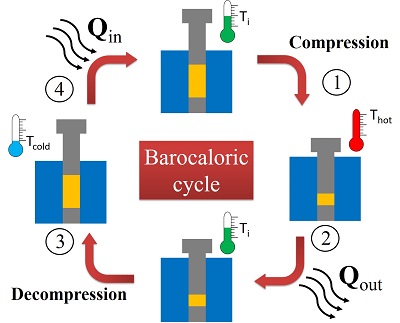
The representation of the barocaloric cycle of a solid material, shown on the side, gives an idea of how vulcanized natural rubber can cool a system, removing heat from it and releasing it to the external environment. In process 1 of the cycle, the rubber (represented by the yellow rectangles) compresses quickly and consequently its temperature increases abruptly (Thot). In process 2, pressure on the rubber is kept constant, but its temperature is reduced by releasing heat to the external environment for thermal equilibrium. Interestingly, in nature two bodies or systems with different temperatures tend to seek thermal equilibrium – the state in which both temperatures are equal. This equilibrium is achieved by transferring heat from the system or hotter body to the cooler one. In process 3 of the cycle, when the rubber reaches its initial temperature (Ti), the pressure rapidly drops, causing the rubber temperature to decrease abruptly (Tcold). In process 4, the external environment transfers heat to the rubber, again for thermal equilibrium. When the rubber reaches the initial temperature, the cycle resumes from a new compression process.
To investigate the barocaloric effect of rubber, Carvalho and the other authors of the paper used samples of vulcanized natural rubber of about 1 cm in diameter. They carried out a systematic study changing the pressure exerted on the samples and their initial temperature and measuring the temperature and entropy variations (both directly related to the heat variation of a system). The experiments were carried out at the Laboratory of i-Caloric Materials (LMiC), one of the thematic laboratories of LNLS, at CNPEM, whose coordinator is Alexandre Carvalho.
After obtaining the experimental measurements of the barocaloric properties of vulcanized natural rubber, the researchers compared them with the results, found in the scientific literature, of other materials with giant or large barocaloric effect. In this comparison, vulcanized natural rubber surpassed all its “competitors.”

The barocaloric effect of vulcanized natural rubber also has advantages with respect to caloric effects generated from the application of a magnetic or electric field, for example – effects that are also studied for solid refrigeration applications. In fact, while relatively low pressures have generated a giant caloric effect on rubber, to produce significant magnetocaloric and electrocaloric effects, very high fields and much more expensive materials than natural rubber are necessary, explains Carvalho.
Besides reporting for the first time in the scientific literature the giant barocaloric effect of vulcanized natural rubber, the paper in ACS Macro Letters contains another important scientific contribution. “The second major contribution is the fact that, for the first time, the effect of the glassy transition of a polymer on the barocaloric effect has been shown,” states Carvalho. The glass transition is a reversible change that occurs in rubber and other materials at a certain temperature. Above the transition temperature, the polymer chains of the rubber acquire more mobility, making the material “rubbery” (more flexible and less hard). Below that temperature, the mobility of the chains decreases and becomes “vitreous” (rigid and relatively brittle). In the ACS Macro Letters article, the authors proposed that the temperature and entropy changes that derive from the compression and decompression of natural rubber are related to the heat generated by the mobility of the polymer chains. Compression of the rubber would lead to a decrease in mobility, which would explain the much lower temperature changes in the vitreous state than in the rubbery state.
As for the application of the discovery, the cooling mechanism based on the barocaloric effect of solid state materials may seem simple, but transferring it to a real device is not easy. “The barocaloric effect on different materials has been studied for several years, but there is still no barocaloric refrigerator prototype patented or described in a paper, as far as we know,” says Carvalho. “Despite the difficulties, we are considering developing a prototype together with researchers from the Department of Mechanical Engineering at the Brazilian State University of Maringá (UEM),” he announces.
History of the work
The idea of the work reported in ACS Macro Letters began in mid-2016 at CNPEM, when the researcher Alexandre Carvalho, the postdoc Nicolau Bom and the student Érik Oda Usuda came across a paper on the elasto-caloric effect of natural rubber (the temperature variation induced by stretching the material), published in Applied Physics Letters. The scientific trio then wondered if an equivalent effect would occur if the rubber were compressed rather than stretched. “More specifically, we wanted to know what would happen in a confined compression,” Carvalho details. They performed the first tests with simple equipment: a pressure cell developed by them and a manual hydraulic press to apply different loads. To prepare the sample, the team used an old school eraser turned into a billet to be fitted into the pressure cell. “The results were encouraging, as we observed that the rubber heated and cooled about 10 degrees from ambient temperature under a relatively low pressure range,” says Carvalho. In early 2017, PhD student William Imamura and postdoc Lucas Soares de Oliveira Paixão joined the group and also devoted their efforts to studying the barocaloric effect of vulcanized natural rubber and other polymers. “We improved our experimental apparatus and our methodology, culminating in the results published in ACS Macro Letters, which will be part of Érik Usuda’s master’s dissertation,” relates Carvalho, who coordinates the LMiC as well as the LNLS XRD1 beamline. In this line, which will be transferred to Sirius (the latest generation synchrotron light source under construction at CNPEM), studies of thermomechanical properties of polymers can be carried out simultaneously with synchrotron radiation analyses, announces Carvalho.
The research was carried out with funding from Brazilian agencies Fapesp, CNPq and Capes, and also LNLS and CNPEM funding.


Manfred Fähnle
Dear Colleague,
I am writing a paper on the collosal barocalorric effect in natural rubber. In former papers phenomenological theoretical explanations are given, based on the Maxwell relation and on the phenomenological Laandau expansion. I want to give an explanation in terms of the statistical physics. I want to calculate the entropy by using Boltzmann’s postulate. To do this, I must calculate the number N of microstates compatibke with the macrostate under the applied hydrostatic pressure. To do this I must know the possible orientations of the monomers of the polymer withrespect to the polymer axis. When applying an exrernal hydrostatic starin, the the macrostate changes and we have other microstates compar´tible with the macrostate under pressure, i.e., the possible arrangements of the angles of the monomers with respect to the polymer axis change, and this gives a change of the entropy. Please tell me the possible orientations .or it woukd be oerfect if you calcukated the numbers N with and without pressure. When you do this then you can of course be coauthor of my paper. Please tell me very soon whether you are interested. Thank you.
Many regards Manfred Fähnle, Max Planck Institute for Intelligent Systems, Stuttgart, Germany