|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|

Perovskites are very promising semiconductor materials for use in solar cells and other optoelectronic devices, but they have a limitation that impairs the useful life of their applications, their low stability. In this context, humidity can be considered the enemy of perovskites, as it is one of the main factors that accelerate the degradation of these materials.
Now, a discovery by researchers from the Brazilian Federal University of ABC (UFABC) brings a very different perspective on the interaction of water with perovskites. With a process based on the simple hydration of three-dimensional perovskite structures, the scientists were able to generate two-dimensional (sheet) and one-dimensional (wire) structures, which, in addition to being more stable than 3D structures, have different properties.
“In controlled amounts, the presence of water can be extremely beneficial for the synthesis of new structures in perovskites”, says André L. M. Freitas, postdoctoral fellow and author, together with Prof. José A. Souza, of the paper that reports this work in the Journal of Materials Chemistry C.
The authors produced micrometric cubes of MAPbBr3, a hybrid material (organic – inorganic) from the perovskite family, which is usually used in the academic environment in solar cells and LEDs. In this material, the inorganic part forms an octahedron, where the central atom (Pb) is surrounded by six halides (Br). “The octahedrons are extremely important in these materials, as they are directly responsible for the physical properties of the material”, explains the postdoc.
To generate the wires, the microcubes were placed in water. To produce the sheets, an organic molecule was added to water. In contact with the liquid, almost instantly, the material lost its crystalline structure, forming what the UFABC duo called an intermediate phase. “We faced a great challenge when analyzing this phase, as the process occurs quickly and many times we had to repeat the experiments to properly characterize it”, says Freitas.
Subsequently, when subjecting the material to drying, the water molecules were expelled from the structure and an interesting conversion took place: the material once again had a perovskite structure, but with a morphology and dimension different from the original ones. “That was one of our main contributions; we were able to control the recrystallization process and obtain different structures and dimensions”, says Freitas.

In the formation of the wires, the process inhibited the growth of the crystalline structure in two directions. In the case of the sheets, the organic molecule added to the water acted as a barrier to the growth of the material in one direction, as if it was a spacer between the layers of octahedra, giving rise to a new structure of the 2D perovskite family. Especially in the 2D structures, this confinement, which occurred on a very small scale, restricted the movement of electrons, resulting in very pronounced quantum effects which modified the electrical and optical properties of the material.
“Our observation revealed that dissolved or degraded material can recrystallize into interesting structures”, summarizes Freitas. According to the authors, by exploring the hydration process of perovskites, the work brings new perspectives to advance the understanding of the degradation and recrystallization of these materials, as well as to understand optical properties such as those involving the dynamics of the exciton (the electron – hole pair generated from the excitation of light in the semiconductor).
The work presents a simple, scalable and sustainable process to produce low-dimensional perovskites with controlled properties. The results could impact both application development and the study of the fundamentals of these versatile materials.
The research received financial support from Brazilian agencies FAPESP and CNPq.
Scientific paper reference: Water-induced dimensionality conversion from 3D perovskites to microwires and 2D hybrid halide perovskites. Andre L. M. Freitas and Jose A. Souza. J. Mater. chem. C, 2023, 11, 6651. https://doi.org/10.1039/D3TC00593C.
Authors contact: joseantonio.souza@ufabc.edu.br, andre_luizmf@yahoo.com.br.
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|

A team of researchers from CeRTEV (one of the largest academic centers for glass research in the world, located in São Carlos, SP, Brazil) has carried out the first experimental observation of the changes that occur in the structure of a glass during relaxation and nucleation, two processes that occur at the nanometer scale in all glasses and that impact their properties.
It is worth remembering that glasses are amorphous materials: their atoms do not appear in an organized and periodic arrangement. Furthermore, they are out of thermodynamic equilibrium and therefore tend to seek stability. In that search, the structure of the glasses undergoes rearrangements, which tend to either make it more fluid (relaxation), or to form the first crystals (nucleation) to, finally, crystallize.
In addition to occurring spontaneously (at the end of almost infinite human times at room temperature), the relaxation and crystallization of glasses can be greatly accelerated by heating the material, which is the method used to produce glass-ceramics. Much more resistant to impacts than common glasses, glass-ceramics have crystalline regions dispersed in the amorphous matrix. Due to their unique properties, they are used in applications such as bulletproof windows and dental restorations.
Understanding the structural changes of glasses during relaxation and nucleation is an old scientific problem, whose resolution was limited by the absence of adequate instrumentation. Therefore, in order to carry out this study, the CeRTEV researchers needed to develop a method. The challenge was finally overcome using experiments based on the nuclear magnetic resonance (NMR) technique combined with computer simulations.
“Our research and the resulting technique offer a valuable tool for monitoring and understanding the relaxation process in many glasses, as well as the early stages of crystal nucleation that occur during heat treatments,” says Henrik Bradtmüller, corresponding author of the article that reports this research in Acta Materialia. “These findings are crucial for the design and production control of technologically advanced glass-ceramics with high performance ”, adds the young German scientist, who has been working as a postdoctoral fellow at UFSCar, with a scholarship of the São Paulo Research Foundation (FAPESP), since 2020.
The discovery
The joint work of highly specialized scientists was one of the keys to achieving success in this research. In fact, the work team added the broad experience of two senior researchers: Professor Edgar Dutra Zanotto (UFSCar) in the area of nucleation and crystallization in glasses, and Professor Hellmut Eckert (IFSC-USP) in the development and refinement of the new NMR technique. Also fundamental were the contributions of postdoctoral fellow Anuraag Gaddam (IFSC-USP), also a FAPESP fellow, who carried out the computational simulations, and Henrik Bradtmüller, who developed and applied the NMR strategies that made the observations possible.

“Through the use of molecular dynamics simulations, we were able to predict the structural changes that occur during glass relaxation,” says Bradtmüller. “In the present contribution we could observe these changes for the first time through sensitive NMR experiments”, he adds. The NMR technique makes it possible to analyze, on the atomic scale, the structure of solid materials, including amorphous structures.
To carry out the experiments, the team chose lithium disilicate (Li2Si2O5), a glass-ceramic widely used, mainly in dental prostheses. The researchers heated it over periods that varied between 15 minutes and 60 days, at 435 °C, a temperature lower than that of the glass transition of this material, in which the atoms gain mobility and the glass begins to become more fluid, without, however, melting.
The samples taken at different times of heating were analyzed using the developed NMR experiments. The results showed, for the first time, what happens to the structure of lithium disilicate during relaxation and nucleation. “The distribution of the network building blocks of this glass (-Si-O-Si-) stays mostly unchanged”, reports Professor Zanotto, who is director of CeRTEV. “In contrast, the network modifier cations (Li+), which are very mobile within the material at annealing temperatures, continously approach a structural configuration that resembles the crystalline state.” Given enough heating time, explains the professor, the first crystal nuclei appear, followed by many others, until the entire material is crystallized.
From now on, the authors of the work hope that the new methodology will be used to study many other vitreous materials and that this detailed understanding of fundamental phenomena will allow adjusting the properties of glass-ceramics to improve their performance and expand their range of applications.
This research was funded by FAPESP.
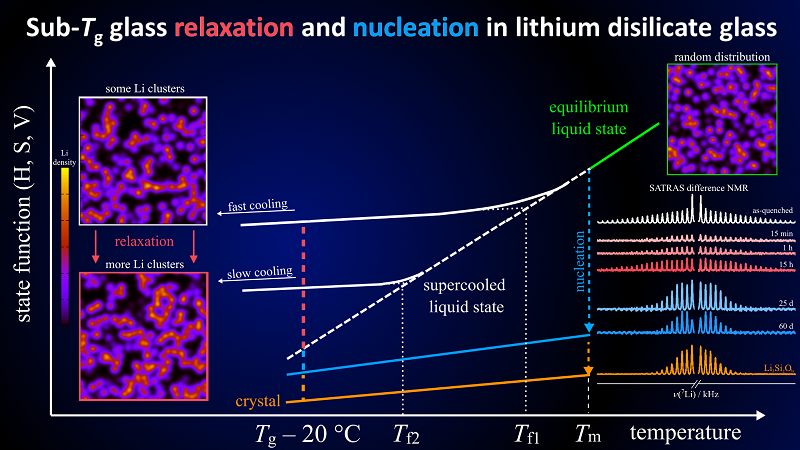
Paper reference: Structural rearrangements during sub-Tg relaxation and nucleation in lithium disilicate glass revealed by a solid-state NMR and MD strategy. Henrik Bradtmüller, Anuraag Gaddam, Hellmut Eckert, Edgar D. Zanotto. Acta Materialia. Volume 240, November 2022, 118318. https://doi.org/10.1016/j.actamat.2022.118318
Author contact: Edgar Dutra Zanotto – dedz@ufscar.br