
Traditional coatings are often effective in protecting steel structures from corrosion by providing a physical barrier that prevents contact between the substrate and corrosive elements. However, when wear generates cracks or pores in these coatings, the substrates are exposed and eventually corrode.
A scientific team from the Brazilian Federal University of Paraná (UFPR) developed a nanotechnology-based coating that offers active protection against corrosion. When the coating shows a flaw, exposing the substrate to corrosive elements, two anti-corrosion mechanisms are activated without human intervention. The coating has very special additives to carry out this intelligent procedure: nanocontainers capable of storing a compound that inhibits corrosion (molybdate) and release it, on demand, in the presence of corrosive elements.
UFPR’s nanocontainers are structures of nanometric dimensions that appear in the form of overlapping two-dimensional sheets (lamellae), separated by a space of a few angstroms. It is in this interlamellar space that molybdate is stored. When a nanoreservoir comes into contact with corrosive environments, such as the sodium chloride (NaCl) solution used in the UFPR experiments, a negative ion exchange reaction occurs: molybdate anions leave the interlamellar space and chloride anions occupy these spaces. In this exchange, the molybdate is free to form a protective film on the exposed substrate surface, while the corrosive anions are “trapped” in the nanoreservoir.
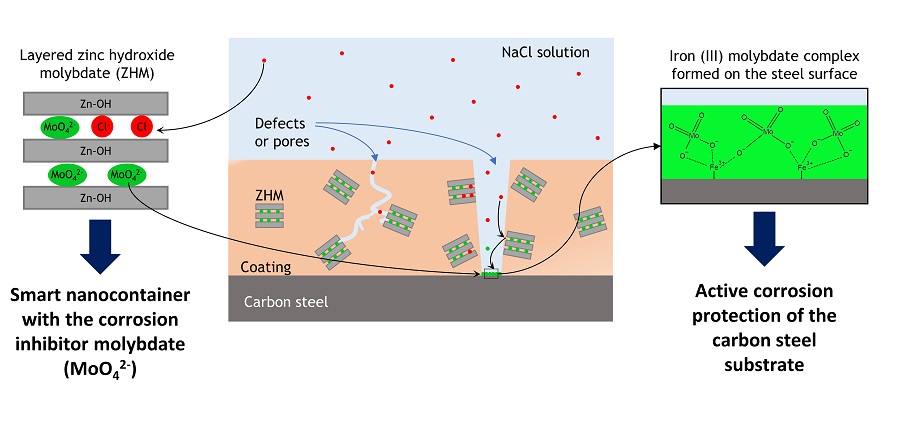
“The main contribution of the work is related to the active corrosion protection of steel. While traditional coatings provide only barrier protection, that is, passive protection, the intelligent coating developed in this work promotes active protection, releasing the molybdate corrosion inhibitor from the lamellar nanoreservoir on demand, through an anion exchange mechanism,” summarizes Débora Abrantes Nunes Leal, doctoral student of the Graduate Program in Materials Engineering and Science at UFPR and corresponding author of the article reporting the research, recently published in ACS Applied Materials & Interfaces. “In addition to releasing the inhibitor in a controlled manner, the lamellar nanoreservoir also “captures” the chloride anions, acting as a trap for these species and, therefore, also contributes to reducing the corrosive environment,” concludes the doctoral student.
Industrial scale use
Produced by means of a quick and simple process and using low-cost, abundant raw materials with low environmental impacts, such as zinc, UFPR’s nanocontainers have the potential for large-scale manufacturing process. When used as coatings additives, as in the UFPR research, nanocontainers could increase the useful life of steel structures found in atmospheres with high concentrations of corrosive compounds, such as, for example, marine atmospheres or close to chemical plants.
According to Débora, there is already a market, mainly in Europe, still incipient, of intelligent coatings to combat corrosion for the aeronautical and petrochemical industries. However, these coatings use as reservoirs of anticorrosive compounds, mainly polymeric microcapsules or double lamellar hydroxides, which are more complex to manufacture, costly and have a greater environmental impact than the lamellar hydroxyses used in UFPR’s nanocontainers . According to the authors of the article, these materials had never been used to store and release corrosion inhibitors before this work, neither in industry nor in the scientific literature.
All the work reported in the article of ACS Applied Materials and Interfaces was carried out by Débora Abrantes Nunes Leal within her still-in-progress doctoral thesis, which started in 2017. The research is guided by Professor Cláudia Marino (advisor), who is an expert in corrosion and protection of materials, and by Professor Fernando Wypych (co-advisor), whose expertise is the synthesis and characterization of lamellar materials. The work received funding from the Brazilian agencies Capes, CNPq and Finep.
Currently, the research on nanocontainers continues, and the authors indicate that they will soon present new results, including those related to the use of other corrosion inhibitors.

[Paper: Zinc-Layered Hydroxide Salt Intercalated with Molybdate Anions as a New Smart Nanocontainer for Active Corrosion Protection of Carbon Steel. Débora Abrantes Leal, Fernando Wypych, and Cláudia Eliana Bruno Marino. ACS Appl. Mater. Interfaces 2020, 12, 17, 19823–19833. https://doi.org/10.1021/acsami.0c02378.]

Tatiane
Parabéns Débora vc é merecedora do seu sucesso!
Débora Leal
Muito obrigada Tatiane!
Ramón S. C. Paredes
Prezada Cláudia, parabéns, seu caminho como pesquisadora da está parada para observar que você está no caminho certo. Bom alunos são orientados por melhores orientadores ainda. Continua assim mesmo, tem muito tempo para o frente para continuar contribuindo com a Ciência. A gente que acredito em você estão orgulhosos de teu sucesso. Grande abraço e estímulos para continuar tu brilhante trabalho como pesquisadora.
Saudosos abraços para ti, Fernando e Debora.
Ramón S. C. Paredes
Débora Leal
Muito obrigada Ramón!!
Rui Eduardo Moreira
Gostaria de parabenizar os autores por este excelente trabalho.
Débora Leal
Muito obrigada Rui!!
Fatima
Parabéns Débora você eleva o nome da nossa cidade. Fico feliz por você e por nós.
É um grande problema esta corrosão principalmente nas construções próximas às praias.
Grande abraço extensivo a seus pais.
Débora Leal
Muito obrigada Fátima! Obrigada pelo carinho e reconhecimento!
A corrosão é um problema recorrente e estamos sempre em busca de novas soluções!
Obrigada! Abraços!!