[PAPER: Bacterial Nanocellulose/MoS2 Hybrid Aerogels as Bifunctional Adsorbent/Photocatalyst Membranes for in-Flow Water Decontamination. Elias P. Ferreira-Neto, Sajjad Ullah, Thais C.A. da Silva, Rafael R. Domeneguetti, Amanda P. Perissinotto, Fábio S. de Vicente, Ubirajara P. Rodrigues-Filho, and Sidney J. L. Ribeiro. ACS Appl. Mater. Interfaces 2020, 12, 37, 41627–41643.]
A team of researchers from Brazilian universities has developed a new material capable of decontaminating water, simultaneously eliminating organic and inorganic pollutants, such as dyes and heavy metals, respectively. The material is in the form of a membrane with the potential to be used as an active filter: as water passes through the membrane, pollutants are adsorbed and degraded. The material can be reused several times, without losing its properties.
The membrane is composed of a three-dimensional network of cellulose nanofibers, coated with molybdenum disulfide (MoS2) nanosheets. Each material performs its function in the filter. Molybdenum disulfide is primarily responsible for adsorbing pollutants and degrading them through photocatalysis [see box]. Nanocellulose operates mainly as support for photocatalysts. Firstly, it allows the construction of an easy to handle macroscopic membrane. In addition, its structure of interwoven nanofibers with a rough surface offers an exceptionally large surface area to contain the photocatalysts. Finally, the flexibility and strength of the nanocellulose allows the membrane to withstand the water flow pressure.
“Although several excellent photocatalysts have been previously developed, one of their disadvantages is the difficult separation and recovery of nanometric materials, thus the idea of producing membranes,” says Elias Ferreira-Neto, postdoctoral fellow at the Laboratory of Photonic Materials, at the Chemistry Institute of UNESP Araraquara. “This study is a first step in the area,” adds Elias, corresponding author of the article reporting on the development of membranes, recently published in Applied Materials & Interfaces (impact factor = 8,758).
Membrane production: from bacterial hydrogel to hybrid aerogel
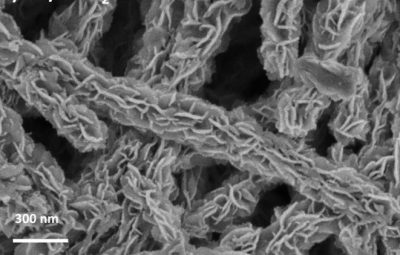
The “recipe” developed by the Brazilian researchers to produce the membranes involves several steps and requires mastering different processes for the synthesis of materials.
In the first step, bacteria from a non-pathogenic strain, placed in an appropriate medium, perform a metabolic process that generates the bacterial nanocellulose hydrogel as a by-product. This highly porous material is composed of 1% of interwoven cellulose nanofibers and 99% of water. The hydrogel is then washed in order to eliminate impurities.
Subsequently, the surface of the nanofibers is coated with controlled structured molybdenum disulfide nanosheets, regularly distributed on the surface of the nanofibers. Finally, this hybrid hydrogel is transformed into aerogel through a drying process, which replaces the pore water with air. The final result is an aerogel membrane composed of bacterial nanocellulose and molybdenum disulfide.
Decontamination assays
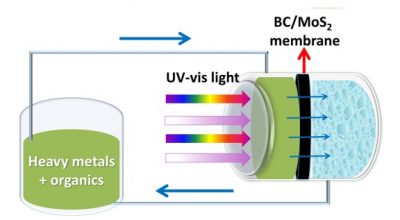
The researchers built a small photoreactor in order to verify the ability of the new membranes to remove the organic and inorganic pollutants in the water. In this device, the contaminated water passes through the membrane, which is illuminated to generate the photocatalytic effect. In the assays, the researchers used an organic pollutant (methylene blue, a compound used as a dye and as a drug) and an inorganic contaminant (hexavalent chromium, a toxic and carcinogenic compound, still used in various industries).
By measuring the presence of contaminants in the water after filtering, the researchers found that the membrane was able to eliminate approximately 96% of the paint and 88% of the heavy metal after 120 minutes of reactor circulation. “The efficiency achieved is in the range of photocatalytic materials of nanoparticulate-like molybdenum disulfide, which is excellent, given that in the supported material, the active surface area exposed to light is much smaller,” says Elias. However, to make the membranes suitable for real applications outside the laboratory, the researchers intend to further increase this efficiency. “The modification of the materials prepared with other photocatalytic nanostructures, such as titanium dioxide (TiO2) and bismuth vanadate (BiVO4), can greatly increase the efficiency of the materials we have already obtained,” says Elias. In addition, the researchers plan to test the action of the membranes against other organic and inorganic compounds that pollute waters, such as drugs, pesticides and other heavy metals.
Aggregated expertise
Entirely carried out in Brazil, more precisely in the state of São Paulo, the work gather the expertise in bacterial cellulose materials from the group led by Professor Sidney Ribeiro (Institute of Chemistry at UNESP Araraquara), and the experience in photocatalysis from the group conducted by Professor Ubirajara Rodrigues Filho (Institute of Chemistry of São Carlos – USP). The research also involved the collaboration of Professor Fábio Simões de Vicente, from the Physics Department of UNESP Rio Claro, to characterize the porosity and textural properties of the materials.
The study is part of Elias Ferreira-Neto’s post-doctoral project, funded by FAPESP. In his doctorate, under the guidance of Professor Ubirajara, and during the research internship abroad conducted with a researcher specialized in aerogels, Elias acquired great experience in the development of inorganic nanoparticles and aerogels for photocatalysis. In these studies, he could identified the great potential of these materials as photocatalysts, as well as their main limitation, the low mechanical resistance and, particularly, the low resistance to capillary pressure in liquid medium.
In the context of Elias’ postdoctoral fellowship, the collaboration between Brazilian research groups could overcome this limitation through the development of hybrid aerogels that combine the excellent mechanical properties of the cellulose support with the photocatalytic and adsorptive properties of the MoS2 nanosheets.

